
B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Landolt-Bornstein: Group II: Volume 6 Molecular Constants from Microwave, Molecular Beam, and Electron Spin Resonance Spectroscopy Springer-Verlag. Please address comments about this page to KH and AM Hellwege (eds.). NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites.įurther, NIST does not endorse any commercial products that may be mentioned on these sites. There may be other web sites that are more appropriate for your purpose. No inferences should be drawn on account of other sites being referenced, or not, from this page. We have provided these links to other web sites because they may have information that would be of interest to you. Such is the case for CO 2, a linear molecule (part (a) in Figure 2.2.8). Electronics An antenna, usually fed from the center, consisting of two equal rods extending outward in a straight line. Chemistry A molecule having two such charges or poles. 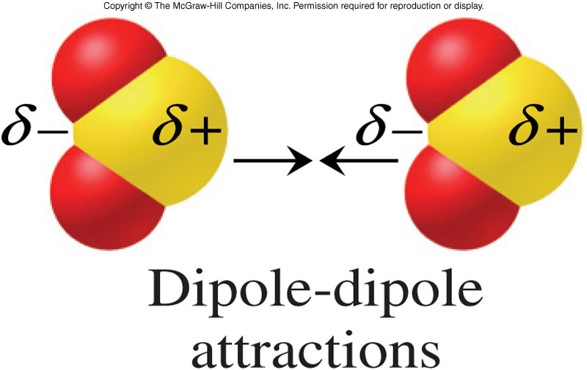
A molecule which has one or more polar covalent bonds may have a dipole moment as a result of the accumulated bond dipoles. Physics A pair of separated electric charges or magnetic poles, of equal magnitude but of opposite sign or polarity. 
If the individual bond dipole moments cancel one another, there is no net dipole moment. One way in which the shapes of molecules manifest themselves experimentally is through molecular dipole moments. But because the molecule is symmetrical, the three dipole arrows cancel and, as a molecule, BH 3 has no net molecular dipole.You are here: Experimental > One molecule all propertiesĮxperimental data for BrF (Bromine monofluoride)īy selecting the following links, you may be leaving NIST webspace. The dipole moment of a molecule is therefore the vector sum of the dipole moments of the individual bonds in the molecule. It is a property of all matter, considering that matter is made up of elementary particles which have an electric charge, namely protons and electrons. The bonds in BH 3 will therefore be somewhat polarized, with the local dipoles oriented towards the hydrogen atoms, as shown below. Polarizability Polarizability usually refers to the tendency of matter, when subjected to an electric field, to acquire an electric dipole moment in proportion to that applied field. A dipole moment is a result of unequal electron distribution, which occurs due to differences in electronegativity between chemically-bonded atoms (since electrons tend to. The electronegativities of boron and hydrogen are 2.04 and 2.20, respectively. Dipole moments occur in any system in which there is a separation of positive and negative electrical charges therefore, they can be found in both ionic and covalent bonds. The BH 3 molecule is planar with all three hydrogens spaced evenly surrounding the boron (trigonal planar). In diatomic molecules, the bond dipole moment determines the. A dipole moment arises in any system wherein there is a separation of charge. 
Consider the molecule boron trihydride (BH 3). The dipole moment measures the extent of net charge separation in the molecule as a whole. Dipole Moment can be defined as the measurement of net polarity in a molecule. Molecules with local dipoles do not necessarily possess a molecular dipole. Because water is asymmetric (it has a bend structure) both of these local dipoles point in the same direction, generating a molecular dipole, in which the entire molecule has a charge imbalance, with the “oxygen end” being anionic and the “hydrogen end” being cationic. We would predict that the H-F bond would be polarized with the greatest electron density towards the fluorine.Ī molecule such as water, with two covalent bonds, will have two local dipoles, each oriented along the covalent bonds, as shown below. A molecular vibration is a periodic motion of the atoms of a molecule relative to each other, such that the center of mass of the molecule remains unchanged. Thus for hydrogen fluoride, the electronegativities are 2.20 and 3.98 for the hydrogen and fluorine, respectively. A dipole arrow is crossed at the beginning (as in a plus sign) and points in the direction of the greatest electron density. Because of this, the polarization of covalent bonds is typically shown using a special arrow (a dipole arrow) to indicate the direction in which the bond is polarized. They must be calculated, however, using sophisticated computer programs, and then rendered in color for visualization. \)Įlectrostatic potential maps are useful because they clearly show the electron distribution around covalent bonds within molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
